Votre question concerne quel type d'offre ?
Votre question concerne quel couloir Ségur ?
Votre question concerne quel dispositif Ségur ?
Votre question concerne quel produit ou service produit?
Votre question concerne quelle thématique ?
To make sure your software complies with the guidelines required to implement INS into software, several documents are available:
Cette réponse vous a-t-elle été utile ?
The cost is determined by the certifying body which grants the certificate.
The daily cost to conduct the audits is at their discretion. The industrial is free to choose which certifying body they want to work with to become certified.
For further information on this topic, we suggest you contact the certifying bodies that have signed a convention agreement with the ANS, listed below:
Cette réponse vous a-t-elle été utile ?
Your application needs to be complete and signed, with all the following documents:
- the MDPH label rules, which describes the labelling process
- the convention regarding the use of the label, which defines the procedures that take place after the label is granted. For example: obligations tied to the label, audit visits, cases of modifying the label, etc.
- the annexes to the convention;
- the license of use for the labelled solution;
- the insurance security plan for the conformity checks relating to the MDPH label;
- if applicable, the decisions issued by partner organisations dealing with exchange fluxes (CNAF/SNGI, Imprimerie Nationale, etc.) to attest you are using the correct one.
The application must be sent in paper AND via electronic mail to:
Agence du Numérique en Santé
Labellisation logiciels Maisons Départementales des Personnes Handicapées
9 rue Georges Pitard 75015 Paris
labellisation.mdph@esante.gouv.fr
Cette réponse vous a-t-elle été utile ?
These documents are made available by the CNDA as a .zip file below.
Cette réponse vous a-t-elle été utile ?
Health professionals are encouraged to use MOS and NOS for three main reasons :
- enhance the interoperability of information systems by harmonising names, attributes, codes and nomenclatures;
- share the same understanding of the information, regardless of which directory or reference document it is taken from;
- facilitate the specification, analysis and conception of a project.
The ANS offers training on MOS-NOS and the elaboration method for exchanges’ functional specs (see our Documentation section).
The illustration below gives you examples of the types of professionals using the MOS-NOS:
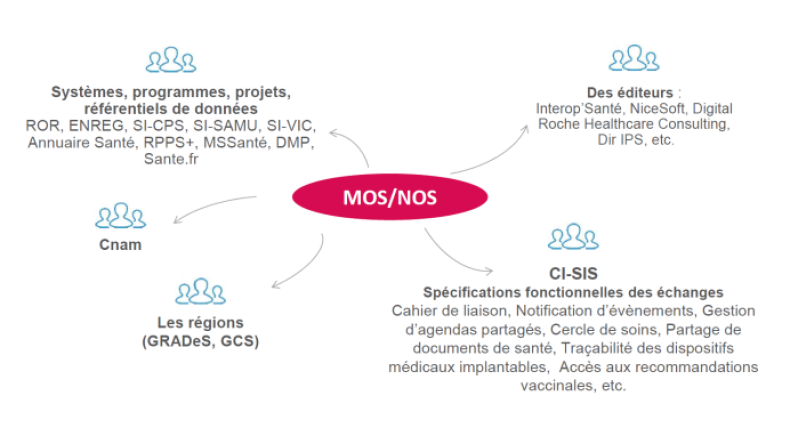
Cette réponse vous a-t-elle été utile ?
Our partner, the GIE Sesam-Vitale, is here to provide you with support and answer your questions about your software project, how to integrate INSi, or solve any potential technical setbacks.
The helpdesk is available by phone at +33 (0)2 43 57 42 88, from 9am to 12noon, and from 2pm to 6pm, Monday to Friday. You may also send an email to centre-de-service@sesam-vitale.fr.
Cette réponse vous a-t-elle été utile ?
In order to make the check lists more transparent and reduce the risk of seeing your Ségur application denied, we have published a document listing common mistakes, which is available below:
Cette réponse vous a-t-elle été utile ?
The MOS is a collection of concepts described homogeneously and neutrally in terms of technologies. It offers a common description of the information processed and exchanged in the information systems to make digital communication easier.
The overall consistency of the MOS is founded on the definition and description of its UML (Unified Modelling Language) concepts.
Some MOS concepts can be coded. They are associated with the nomenclatures of health objects (NOS), i.e. lists of codes/labels.
You are encouraged to use MOS and NOS to :
- optimise and coordinate efforts when you analyse or conceive a system (or an application) by re-using the same semantic components;
- make sure there is consistency in your internal developments and with external systems, for the best possible interoperability.
Cette réponse vous a-t-elle été utile ?
The European Commission’s studies have determined there is a need for a base vocabulary, such as MOS and NOS, which can be used as a starting point for:
- develop and evolve information systems (IS) to formalise the conceptual and logical data they utilise (for instance, the MOS is a reference for the modelling of the RPPS);
- share information between ISs to create specific models of data they can use (thus, the MOS is underlying the ROR exposure model);
- combine and synthetise elements originating from different sources;
- publish data in a common format, such as a directory or catalogue of service (for example, the Annuaire Santé / Health Pro Directory).
Cette réponse vous a-t-elle été utile ?
Upgrading your products allows you to offer your customers a solution that complies with current regulations. Failure to meet this requirement will have several impacts:
-
Your customers will not incorporate the INS into their software, which will entail possible penalties with regard to several systems (Ségur Wave 1, HOPEN, portal listing in Mon Espace Santé, etc.) ;
-
Your customers will not be in compliance with the law. They will therefore be responsible for any errors in the identification of their patients and users ;
-
Sharing health data by your customers will be more time consuming ;
-
The identity used by your customers may not be the right one, for instance if a patient uses their married name or if the patient is a child registered under their parent’s social security number.
Cette réponse vous a-t-elle été utile ?
The application for the SI-MDPH label is open to any legal entity with a software solution aimed at the departmental disability centres (MDPHs). This includes the MDPHs themselves, which develop their own solutions in conjunction with the relevant Departmental Council.
The label application is a voluntary process: Anyone can be a candidate if they feel that their solution complies with the current requirements and the scope of the sector’s functional standards.
Cette réponse vous a-t-elle été utile ?
Yes, you need to have the ISO 9001 or the ISO 13485 certification to be eligible. You may begin the QHN certification process while you are in the course of making an application for/or renewing these ISO 9001 or the ISO 13485 certifications.
Cette réponse vous a-t-elle été utile ?
Yes, as long as the industrial remains responsible for the compliance of these processes with the requirements set out to obtain the QHN certification – regardless of hired external contractors or if some of the components are published or made by third-party companies.
Indeed, in the whole production system’s organisation, the industrial is the direct contact with a health professional or structure, and must make sure each requirement set out in the “Référentiel Qualité Hôpital Numérique” (QHN Referential, available in our Documentation section) is met.
Cette réponse vous a-t-elle été utile ?
A base product (“Produit socle”) is a solution, a service, or device sold by an eHealth industrial, which is aimed at fulfilling the needs of a particular activity, practice or profession. It is not bound to the purchase of a previous product. It may be completed with Option Products (“Produit option”) to extend its functional coverage.
An option product (“Produit option”) is a solution, service, or device sold by an eHealth industrial to meet specific needs extending the functional perimeter of one or several base products (“produits socle”) to which it adds on. This means a base product must be purchased, prior to the use of an option product.
In the case of a base product being offered as an option to another product, we suggest you first declare the product as a “produit socle”. Then you will be able, from this entry, to duplicate and convert the product as a “produit option”.
Cette réponse vous a-t-elle été utile ?


