Votre question concerne quel type d'offre ?
Votre question concerne quel couloir Ségur ?
Votre question concerne quel dispositif Ségur ?
Votre question concerne quel produit ou service produit?
Votre question concerne quelle thématique ?
There are two steps to follow:
Step 1: Administrative phase
Should you wish to order test CPS cards or register authorisations for test software, you may:
- read our complete products offer ;
- place an order
To place an order, select your Profile and your Structure, then choose “Produits de développement. Commander des produits de développement : carte et/ou certificat logiciel de test" (Test products for products, cards and/or test software).
Step 2: Technical phase
Use the card you validated after step 1 in order to connect with the Trusted Platform IGC-Santé. You will be able to order, withdraw, monitor and revoke test certificates through the IHM or Webservice interface.
In order to do so, make sur you have inserted your test card in the card reader.
Read more about specific setup guidelines:
Cette réponse vous a-t-elle été utile ?
In an inclusive way, all digital services aimed at citizens and useful for their healthcare can apply. Eligible services must be for the use of patients/citizens and proposed by public or private actors in the medical (health, wellness), social-medical, or social fields. The services must be included the definition set out by the L. 1111-13-1 bill:
These services may include mobile apps, websites, connected objects, medical devices. They may be free services or not.
Services in the realm of the EU regulation 2017/745 relating to medical devices must be conform to security and performance requirements. If your solution passes the conformity requirements, it will receive the CE label for medical devices.
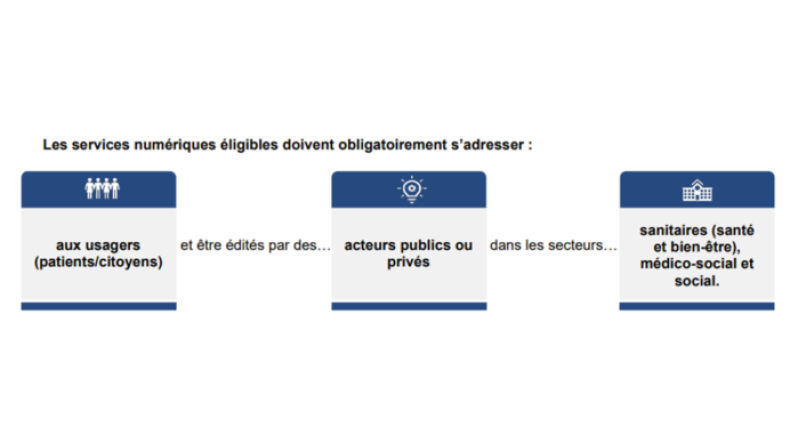
Cette réponse vous a-t-elle été utile ?
All the resources for funding are available in our Ségur section for digital equipment.
Cette réponse vous a-t-elle été utile ?
For your application to be complete, you need to include several credentials and documents.
The list of documents and credentials needed is detailed in the Application form (download it below). You will need to send and application for each product you wish to label.
You must return your application form with all the supporting material by email AND via postal services:
- the complete application form needs to be sent by mail to:
Agence du numérique en santé (ANS)
Labellisation logiciel Maisons et Centres de Santé
9, rue Georges Pitard
75015 Paris - follow up with sending digital versions (PDF scans) of the agreement and its annexes, in two copies, signed and completed to:
Cette réponse vous a-t-elle été utile ?
The IGC-Santé is dedicated to the health sector and follows strict procedures in terms of data collection, professional identification, and works with certified authorities (RPPS register, etc.).
The certificates issued by the IGC guarantee the security of software or electronic cards, such as the CPS card.
The IGC also manages the publication of these certificates and can revoke them – this is signalled to the apps using certificates in revocation listings.
Cette réponse vous a-t-elle été utile ?
There are two main reasons for the creation of IGC-Santé :
- guaranteeing the security of private keys and certificates issued by the ANS: the access to these private keys must be limited in order to prevent duplicates or their installation more than one device;
- maintaining a continuity in services: many health apps used to work on certificates issued by former CKI that ceased their activity in January 2021. These apps must be compatible with certificates issued by the IGC-Santé.
In addition, these certificates meet the security standards (risk analysis, safety policies), or “Certifying Policies” that comply with the PGSS-IS guidelines.
Cette réponse vous a-t-elle été utile ?
When your software gets the label attribution, the successful candidate will receive by email the visual elements you will need to communicate about it.
The editor of the solution (or the solution range) will receive the label after signing the agreement with the ANS. They will be able to use the label on commercial publications, technical documents, with no particular restriction on the type of format used (paper, Internet, intranet, etc.)
The recipient must mention the level of the label (1 or 2) they obtained. These specifications are set out in the Functional referential (in our Documentation section)
Cette réponse vous a-t-elle été utile ?
In order for your service to be referenced in the My Health Space database, it must comply with the eHealth doctrine.
The doctrine is re-edited each year and defines the base rules of good practice for security, interoperability, and ethics applicable to eHealth services.
Cette réponse vous a-t-elle été utile ?
My Health Space includes the following health data:
- The medical profile entered by the patient, in an unstructured format (preconditions, allergies);
- Health measurements (weight, height, BMI, temperature, waist size, heart rate, blood pressure, glycemic index, level of pain) in FHIR format;
- Health documents (for example: test and diagnosis results, hospitalisation reports, medical treatments).
Cette réponse vous a-t-elle été utile ?


