Votre question concerne quel type d'offre ?
Votre question concerne quel couloir Ségur ?
Votre question concerne quel dispositif Ségur ?
Votre question concerne quel produit ou service produit?
Votre question concerne quelle thématique ?
There are two steps to follow:
Step 1: Administrative phase
Should you wish to order test CPS cards or register authorisations for test software, you may:
- read our complete products offer ;
- place an order
To place an order, select your Profile and your Structure, then choose “Produits de développement. Commander des produits de développement : carte et/ou certificat logiciel de test" (Test products for products, cards and/or test software).
Step 2: Technical phase
Use the card you validated after step 1 in order to connect with the Trusted Platform IGC-Santé. You will be able to order, withdraw, monitor and revoke test certificates through the IHM or Webservice interface.
In order to do so, make sur you have inserted your test card in the card reader.
Read more about specific setup guidelines:
Cette réponse vous a-t-elle été utile ?
To register on the Convergence platform, you need to download the account request form to open an account as a company representative.
Before completing this form, please check that your company is not already represented by another person who is able to create an account for you to access the Platform. If you have any questions, please contact support at ans-support-convergence@esante.gouv.fr.
As soon as you receive the e-mail confirming the creation of your account, you will be able to initialize your password and access the Platform after accepting the General Terms of Use.
Cette réponse vous a-t-elle été utile ?
Health professionals are encouraged to use MOS and NOS for three main reasons :
- enhance the interoperability of information systems by harmonising names, attributes, codes and nomenclatures;
- share the same understanding of the information, regardless of which directory or reference document it is taken from;
- facilitate the specification, analysis and conception of a project.
The ANS offers training on MOS-NOS and the elaboration method for exchanges’ functional specs (see our Documentation section).
The illustration below gives you examples of the types of professionals using the MOS-NOS:
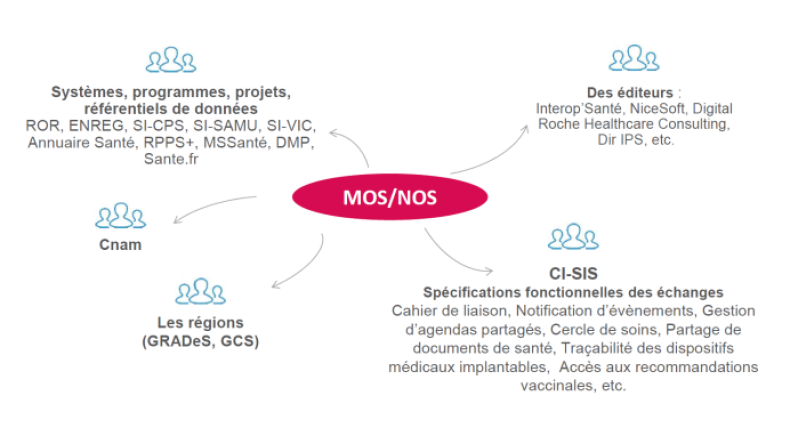
Cette réponse vous a-t-elle été utile ?
The IGC-Santé is dedicated to the health sector and follows strict procedures in terms of data collection, professional identification, and works with certified authorities (RPPS register, etc.).
The certificates issued by the IGC guarantee the security of software or electronic cards, such as the CPS card.
The IGC also manages the publication of these certificates and can revoke them – this is signalled to the apps using certificates in revocation listings.
Cette réponse vous a-t-elle été utile ?
The MOS is a collection of concepts described homogeneously and neutrally in terms of technologies. It offers a common description of the information processed and exchanged in the information systems to make digital communication easier.
The overall consistency of the MOS is founded on the definition and description of its UML (Unified Modelling Language) concepts.
Some MOS concepts can be coded. They are associated with the nomenclatures of health objects (NOS), i.e. lists of codes/labels.
You are encouraged to use MOS and NOS to :
- optimise and coordinate efforts when you analyse or conceive a system (or an application) by re-using the same semantic components;
- make sure there is consistency in your internal developments and with external systems, for the best possible interoperability.
Cette réponse vous a-t-elle été utile ?
The European Commission’s studies have determined there is a need for a base vocabulary, such as MOS and NOS, which can be used as a starting point for:
- develop and evolve information systems (IS) to formalise the conceptual and logical data they utilise (for instance, the MOS is a reference for the modelling of the RPPS);
- share information between ISs to create specific models of data they can use (thus, the MOS is underlying the ROR exposure model);
- combine and synthetise elements originating from different sources;
- publish data in a common format, such as a directory or catalogue of service (for example, the Annuaire Santé / Health Pro Directory).
Cette réponse vous a-t-elle été utile ?
There are two main reasons for the creation of IGC-Santé :
- guaranteeing the security of private keys and certificates issued by the ANS: the access to these private keys must be limited in order to prevent duplicates or their installation more than one device;
- maintaining a continuity in services: many health apps used to work on certificates issued by former CKI that ceased their activity in January 2021. These apps must be compatible with certificates issued by the IGC-Santé.
In addition, these certificates meet the security standards (risk analysis, safety policies), or “Certifying Policies” that comply with the PGSS-IS guidelines.
Cette réponse vous a-t-elle été utile ?
You may divide your service into as many products as you wish to register on the platform. The appropriate segmentation must correspond to your activity and the roadmaps/processes available on the platform.
It is important to include all the different versions associated with support or devices as different products, as they could impact the maturity level assessment of your solution.
In order to make the identification of products easier, each edition and version of the commercialised products needs to be registered. We suggest you use this format to name your product - (Name of the Product) (OS) (Device) - with the following terms:
- device can be named “Mobile” or “Website”
- OS can be named “iOS”, “Android”, “Windows”, “Linux” or any other operating system.
In addition, for registration into Mon Espace Santé (My Health Space): you must add as a product the solution that will be visible to the citizen/user in the service catalogue. The commercial name must be the same as the name visible by the citizen.
Cette réponse vous a-t-elle été utile ?
The Convergence platform supports you in developing your business and defining your plan for compliance with regulations specific to the e-health sector.
To this end, the platform is organized around two tracks:
- digital health doctrine ;
- My health space.
Each of these paths is broken down into steps:
- doctrine is divided into 4 steps: Evaluation and projection socles , Teleconsultation, teleexpertise, telecare;
- My Health Space is divided into 2 steps: referencing without data exchange and referencing with data exchange.
The procedures include questionnaires which, once completed, will enable you to assess the maturity and/or compliance of your products with the digital health doctrine, or the assessments required for My Health Space registration.
Cette réponse vous a-t-elle été utile ?
The first step in completing the platform is to identify a manager who will oversee the completion operations. This person can then make an inventory of the products to be included in the scope of data entry, and list the resources to be mobilized: product managers, technical and functional experts, etc. Despite efforts to contextualize the questions, knowledge of healthcare IS interoperability, security and the ambitions of the doctrine of digital health is a prerequisite.
The questionnaires can then be read in groups. Based on the completion of an initial product, the reading of the questionnaires enables the identification of subjects that have been mastered, as well as those that require further investigation.
The pilot can then propose regular meetings with all product managers to monitor the progress of data entry, guarantee the consistency of responses and share expert feedback.
Cette réponse vous a-t-elle été utile ?
There are two main user profiles on the Convergence platform:
- Manager: this is either the Corporate Officer or a person authorized by the Corporate Officer; he/she is authorized to add new contributors, create products and grant rights to contributors from his/her company;
- Contributor: authorized to create accounts for other contributors, create products and complete questionnaires, within the limits of the rights assigned to him/her.
Contributors are given access rights to questionnaires for products already created within the platform, and to questionnaires relating to global strategy.
Contributors who do not have the right to validate questionnaires can only save drafts for questionnaires to which they have access.
Cette réponse vous a-t-elle été utile ?


