Votre question concerne quel type d'offre ?
Votre question concerne quel couloir Ségur ?
Votre question concerne quel dispositif Ségur ?
Votre question concerne quel produit ou service produit?
Votre question concerne quelle thématique ?
To make sure your software complies with the guidelines required to implement INS into software, several documents are available:
Cette réponse vous a-t-elle été utile ?
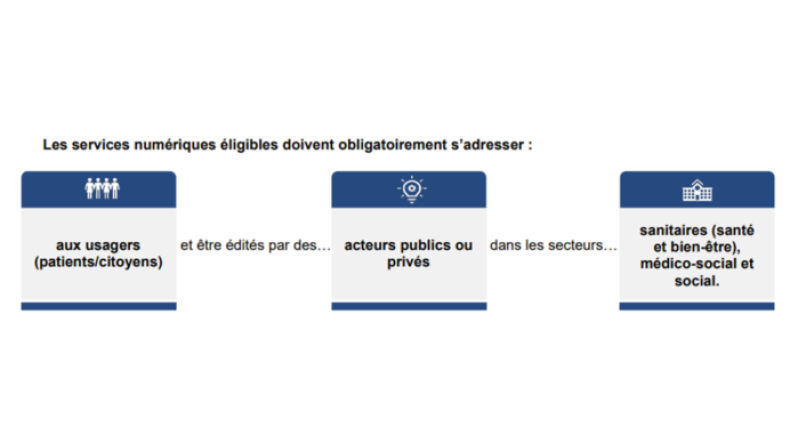
In an inclusive way, all digital services aimed at citizens and useful for their healthcare can apply. Eligible services must be for the use of patients/citizens and proposed by public or private actors in the medical (health, wellness), social-medical, or social fields. The services must be included the definition set out by the L. 1111-13-1 bill:
These services may include mobile apps, websites, connected objects, medical devices. They may be free services or not.
Services in the realm of the EU regulation 2017/745 relating to medical devices must be conform to security and performance requirements. If your solution passes the conformity requirements, it will receive the CE label for medical devices.
Cette réponse vous a-t-elle été utile ?
Our partner, the GIE Sesam-Vitale, is here to provide you with support and answer your questions about your software project, how to integrate INSi, or solve any potential technical setbacks.
The helpdesk is available by phone at +33 (0)2 43 57 42 88, from 9am to 12noon, and from 2pm to 6pm, Monday to Friday. You may also send an email to centre-de-service@sesam-vitale.fr.
Cette réponse vous a-t-elle été utile ?
In order to make the check lists more transparent and reduce the risk of seeing your Ségur application denied, we have published a document listing common mistakes, which is available below:
Cette réponse vous a-t-elle été utile ?
A software publisher usually integrates several terminologies that cover standard usages: common names and codes, language names and codes, countries...
Then they integrate “Base Terminologies” (drug terminologies, billing, administrative) and occupation-specific terminologies (Terminology of the living, cardiology terminology, geriatric terminologies, etc.).
Cette réponse vous a-t-elle été utile ?
Generally speaking, the PGSSI-S needs to be applied as soon as personal health data are being handled. It is relevant to the public sector as well as the private sector, health professionals, workers of the social-health and social sectors, healthcare establishments and service providers.
As a patient, the PGSSI-S is a seal of guarantee on the accountability of digital health ecosystems.
Cette réponse vous a-t-elle été utile ?
Complying with the PGSSI-S frames of reference is either required by law (if the documents have been approved by a ministerial decree) or meant to be followed on a short-term basis until the documents are approved by the ministry.
Cette réponse vous a-t-elle été utile ?
Upgrading your products allows you to offer your customers a solution that complies with current regulations. Failure to meet this requirement will have several impacts:
-
Your customers will not incorporate the INS into their software, which will entail possible penalties with regard to several systems (Ségur Wave 1, HOPEN, portal listing in Mon Espace Santé, etc.) ;
-
Your customers will not be in compliance with the law. They will therefore be responsible for any errors in the identification of their patients and users ;
-
Sharing health data by your customers will be more time consuming ;
-
The identity used by your customers may not be the right one, for instance if a patient uses their married name or if the patient is a child registered under their parent’s social security number.
Cette réponse vous a-t-elle été utile ?
A base product (“Produit socle”) is a solution, a service, or device sold by an eHealth industrial, which is aimed at fulfilling the needs of a particular activity, practice or profession. It is not bound to the purchase of a previous product. It may be completed with Option Products (“Produit option”) to extend its functional coverage.
An option product (“Produit option”) is a solution, service, or device sold by an eHealth industrial to meet specific needs extending the functional perimeter of one or several base products (“produits socle”) to which it adds on. This means a base product must be purchased, prior to the use of an option product.
In the case of a base product being offered as an option to another product, we suggest you first declare the product as a “produit socle”. Then you will be able, from this entry, to duplicate and convert the product as a “produit option”.
Cette réponse vous a-t-elle été utile ?
In order for your service to be referenced in the My Health Space database, it must comply with the eHealth doctrine.
The doctrine is re-edited each year and defines the base rules of good practice for security, interoperability, and ethics applicable to eHealth services.
Cette réponse vous a-t-elle été utile ?
My Health Space includes the following health data:
- The medical profile entered by the patient, in an unstructured format (preconditions, allergies);
- Health measurements (weight, height, BMI, temperature, waist size, heart rate, blood pressure, glycemic index, level of pain) in FHIR format;
- Health documents (for example: test and diagnosis results, hospitalisation reports, medical treatments).
Cette réponse vous a-t-elle été utile ?
The State evaluates the following criteria:
- Terminology conceived by one of the ANS’ Production Units
- Terminology conceived by a Production Unit with a convention
- Terminology in usage
- Terminology compatible with the ANS right imposition
- Reference terminology
- “Open” terminology
- Maturity of the Production Unit in charge of the terminology’s conception and maintenance
- Local terminology sets (“small terminologies”)
- Terminology conceived by a “fragile” Production Unit / Terminology conceived by a Production Unit without a distribution point
Cette réponse vous a-t-elle été utile ?


