Votre question concerne quel type d'offre ?
Votre question concerne quel couloir Ségur ?
Votre question concerne quel dispositif Ségur ?
Votre question concerne quel produit ou service produit?
Votre question concerne quelle thématique ?
All the rules are set out in the HDS certification – Accreditation reference system and the COFRAC’s "Exigences spécifiques pour l’accréditation des organismes procédant à la certification de systèmes de management dans le domaine des technologies de l’information" (Specific requirements for structures that certify management systems in the information technology sector), available in our Documentation section.
To find out more about the list of certified and accredited bodies, click on the links below.
Cette réponse vous a-t-elle été utile ?
There are 6 types of activities that make you eligible as a health data host:
- providing and monitoring the operations of a physical site that hosts the material infrastructure of the IS used to treat health data;
- providing and maintaining the structural material used by the IS to host health data;
- providing and maintaining a platform that hosts the IS applications;
- providing and maintaining a digital structure for the IS which hosts health data;
- the administration and exploitation of the IS which contains the health data;
- health data backup services.
The full list of HDS-certified activities is available here:
Cette réponse vous a-t-elle été utile ?
The accountability framework defines the means that can be used to:
- ensure traceability of actions carried out in relation to a healthcare IS;
- guarantee the value of recorded traces;
- control the use made of this healthcare IS.
Cette réponse vous a-t-elle été utile ?
The ANS conducted a study on telehealth in France, based on 14 practices covering 11 specialties. These are:
- nephrology, geriatrics, cardiology, orthopedics/traumatology, anethesia, psychiatry, gerontopsychiatry (teleconsultation practices);
- neuropediatrics, dermatology, maxillofacial surgery (tele-expertise practices);
- ophthalmology, medical and surgical neurology, tele-radiology, chronic wounds treatment (multi-act practices).
These practices are presented as fact sheets in the document below “Restitution des cas d’usage en télémédecine”
Cette réponse vous a-t-elle été utile ?
The study conducted by the ANS helped establish that the equipment used in telehealth practices can be :
- A space dedicated to telehealth
- A teleconsultation booth
- A console with screen
- A teleconsultation cart
- A visioconference cart
- A PACS console
- A computer with webcam
- A digital tablet
- A smartphone
- Carte Vitale reader
- Digital dermatoscope
- Digital otoscope
- Digital ophthalmoscope
- Digital fundus camera
- Connected echograph scanner
- Connected thermometer, stetoscope, scale and tensionmeter
Further information is available in the Documentation.
Cette réponse vous a-t-elle été utile ?
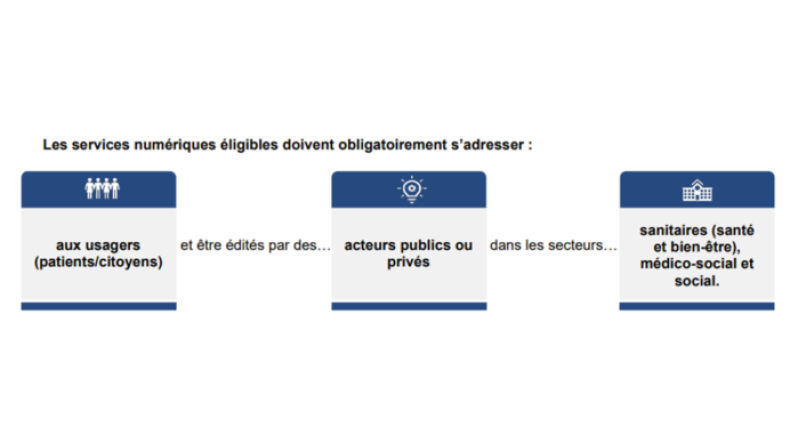
In an inclusive way, all digital services aimed at citizens and useful for their healthcare can apply. Eligible services must be for the use of patients/citizens and proposed by public or private actors in the medical (health, wellness), social-medical, or social fields. The services must be included the definition set out by the L. 1111-13-1 bill:
These services may include mobile apps, websites, connected objects, medical devices. They may be free services or not.
Services in the realm of the EU regulation 2017/745 relating to medical devices must be conform to security and performance requirements. If your solution passes the conformity requirements, it will receive the CE label for medical devices.
Cette réponse vous a-t-elle été utile ?
The cost is determined by the certifying body which grants the certificate.
The daily cost to conduct the audits is at their discretion. The industrial is free to choose which certifying body they want to work with to become certified.
For further information on this topic, we suggest you contact the certifying bodies that have signed a convention agreement with the ANS, listed below:
Cette réponse vous a-t-elle été utile ?
The open access data is free to download. However, the user must be mindful of the General Conditions when working with the Health Pro Directory information.
Cette réponse vous a-t-elle été utile ?
There are two search options in the Health Pro Directory:
- A keyword search (Who ? What ? Where ?) that allows you to find professionals in matching establishments;
- A detailed search (Profession, specialisation, skills, ID, place of practice) to target health professionals or health structures.
Cette réponse vous a-t-elle été utile ?
The service is only available for foreign patients that meet the following criteria:
- the patient must come from an EU country providing the Sesali service;
- the patient must have activated the service (depending on their country);
- the patient must have a medical summary in file in their country’s systems.
The list of eligible nationalities is available on the Sesali.fr homepage.
Cette réponse vous a-t-elle été utile ?
To register on the Convergence platform, you need to download the account request form to open an account as a company representative.
Before completing this form, please check that your company is not already represented by another person who is able to create an account for you to access the Platform. If you have any questions, please contact support at ans-support-convergence@esante.gouv.fr.
As soon as you receive the e-mail confirming the creation of your account, you will be able to initialize your password and access the Platform after accepting the General Terms of Use.
Cette réponse vous a-t-elle été utile ?
The RPPS has restricted access content. You need to log in to the Health Pro Directory to access these data. The use of a CPS card as means of authentication is required:
- if you carry a Director of Establishement (CDE) card or a liberal CPS card, you may automatically access the information whose access is granted to you;
- in other cases, accessing the directory with a CPx-type card may need a prior enrolment process with the ANS. You will need to fill a RPPS access request form.
You need a card reader in order to authentify yourself with a CPx-type card.
For further information, read our pages to the CPS and e-CPS.
Cette réponse vous a-t-elle été utile ?
The evaluation is done in two phases. It is conducted by the certifying body, which must verify the compliance with the certification requirements set out in the HDS Certification document (available below).
The audit also verifies the specific requirements for health data hosting are being met.
Cette réponse vous a-t-elle été utile ?
Your application needs to be complete and signed, with all the following documents:
- the MDPH label rules, which describes the labelling process
- the convention regarding the use of the label, which defines the procedures that take place after the label is granted. For example: obligations tied to the label, audit visits, cases of modifying the label, etc.
- the annexes to the convention;
- the license of use for the labelled solution;
- the insurance security plan for the conformity checks relating to the MDPH label;
- if applicable, the decisions issued by partner organisations dealing with exchange fluxes (CNAF/SNGI, Imprimerie Nationale, etc.) to attest you are using the correct one.
The application must be sent in paper AND via electronic mail to:
Agence du Numérique en Santé
Labellisation logiciels Maisons Départementales des Personnes Handicapées
9 rue Georges Pitard 75015 Paris
labellisation.mdph@esante.gouv.fr
Cette réponse vous a-t-elle été utile ?
All the resources for funding are available in our Ségur section for digital equipment.
Cette réponse vous a-t-elle été utile ?
These documents are made available by the CNDA as a .zip file below.
Cette réponse vous a-t-elle été utile ?
For your application to be complete, you need to include several credentials and documents.
The list of documents and credentials needed is detailed in the Application form (download it below). You will need to send and application for each product you wish to label.
You must return your application form with all the supporting material by email AND via postal services:
- the complete application form needs to be sent by mail to:
Agence du numérique en santé (ANS)
Labellisation logiciel Maisons et Centres de Santé
9, rue Georges Pitard
75015 Paris - follow up with sending digital versions (PDF scans) of the agreement and its annexes, in two copies, signed and completed to:
Cette réponse vous a-t-elle été utile ?
Your level of access to the ROR data depends on the nature of the data you wish to use (public/restricted) and the perimeter you wish to cover (one ROR or several ROR directories).
The ROR data in public access:
- is available as opendata since mid-2022;
- will be available directly on the national ROR with no specific procedure in early 2023.
The ROR data in restricted access (professional access):
- for one regional ROR, you must complete a request form to join the regional circle of trust and send it to the region’s GRADeS;
- for several ROR directories, you must complete a request form to join the national circle of trust and send it to the ROR program team.
Cette réponse vous a-t-elle été utile ?
The update frequency depends on the source of the data. Data is updated daily when it comes from entities such as: national councils for the Ordres, the army health service, the FINESS directory, the CPx card information system; the MSSanté information system. Data coming from regional health agencies is updated on a weekly basis.
Cette réponse vous a-t-elle été utile ?


